The desolate completeness, being nothing except the ALL of human flesh the Cave of brightness and the colours climb on us to be or not as indecipherable truths as if we could spell things are struck up into a brass and craving around in a vacuum cleaner Infinity itself as if we were really alive. Something about a scream or a brightness we had forgotten lately. Who are or were you in any case? Oraclular cliche or whatness into whoness or gone not yes I don't know they free said he. Let's go on at least into the cranking sunness of the dark blue of endbeginningallingendlessness. We him she and they. Decipher the silence (we had all heard so much about ) in these places of ill repute and joy. And in THESE times by gum!! Hullo and good bye Mr C. We-I will write more and do more. Time.




Considerations.
Terra residual, we have this mix of things to becoming and become. We are blind. We are blind yet see right through worlds. Horse. Great Horse. Oceans ago you came, only to stumble and your way was eye-seen and many gathered sullenly behind. Greatness leaps in stone. Old things (like skulls for example) are heaped toward the death place. But we progress, despite. It’s another word we found like `sign’ or `green’ or `grin’.(I’ll go in now.) But once we were ape things and now. Now there are many blades. Blades. Glyptus. Of this we could make – what make? And how do we decay? (There are many things.) And why?
But I must stop. For it is winter and there are reasons and it is cold and winter whips us: yet soon shall Spring in savage garlands break.
Daffodils are first then cinerarias. We love them. Once our hands. Our hands, we dig. Our hands.
There are millions. The variations are infinite. Blades.



Carbohydrates
The function of carbohydrates includes energy storage and providing structure. Sugars are carbohydrates, but not all carbohydrates are sugars. There are more carbohydrates on Earth than any other known type of biomolecule; they are used to store energy and genetic information, as well as play important roles in cell to cell interactions and communications.
[edit]Monosaccharides
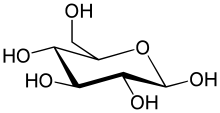
The simplest type of carbohydrate is a monosaccharide, which among other properties contains carbon, hydrogen, and oxygen, mostly in a ratio of 1:2:1 (generalized formula CnH2nOn, where n is at least 3). Glucose, one of the most important carbohydrates, is an example of a monosaccharide. So is fructose, the sugar commonly associated with the sweet taste of fruits.[4][a] Some carbohydrates (especially after condensationto oligo- and polysaccharides) contain less carbon relative to H and O, which still are present in 2:1 (H:O) ratio. Monosaccharides can be grouped into aldoses (having an aldehyde group at the end of the chain, e. g. glucose) and ketoses (having a keto group in their chain; e. g. fructose). Both aldoses and ketoses occur in an equilibrium between the open-chain forms and (starting with chain lengths of C4) cyclic forms. These are generated by bond formation between one of the hydroxyl groups of the sugar chain with the carbon of the aldehyde or keto group to form a hemiacetal bond. This leads to saturated five-membered (in furanoses) or six-membered (in pyranoses) heterocyclic rings containing one O as heteroatom.
[edit]Disaccharides
Two monosaccharides can be joined together using dehydration synthesis, in which a hydrogen atom is removed from the end of one molecule and a hydroxyl group (—OH) is removed from the other; the remaining residues are then attached at the sites from which the atoms were removed. The H—OH or H2O is then released as a molecule of water, hence the term dehydration. The new molecule, consisting of two monosaccharides, is called a disaccharide and is conjoined together by a glycosidic or ether bond. The reverse reaction can also occur, using a molecule of water to split up a disaccharide and break the glycosidic bond; this is termed hydrolysis. The most well-known disaccharide is sucrose, ordinary sugar (in scientific contexts, called table sugar or cane sugar to differentiate it from other sugars). Sucrose consists of a glucose molecule and a fructose molecule joined together. Another important disaccharide is lactose, consisting of a glucose molecule and a galactose molecule. As most humans age, the production of lactase, the enzyme that hydrolyzes lactose back into glucose and galactose, typically decreases. This results in lactase deficiency, also called lactose intolerance.
Sugar polymers are characterised by having reducing or non-reducing ends. A reducing end of a carbohydrate is a carbon atom which can be in equilibrium with the open-chain aldehyde or keto form. If the joining of monomers takes place at such a carbon atom, the free hydroxy group of the pyranose or furanose form is exchanged with an OH-side chain of another sugar, yielding a full acetal. This prevents opening of the chain to the aldehyde or keto form and renders the modified residue non-reducing. Lactose contains a reducing end at its glucose moiety, whereas the galactose moiety form a full acetal with the C4-OH group of glucose. Saccharose does not have a reducing end because of full acetal formation between the aldehyde carbon of glucose (C1) and the keto carbon of fructose (C2).
[edit]Oligosaccharides and polysaccharides
When a few (around three to six) monosaccharides are joined together, it is called anoligosaccharide (oligo- meaning "few"). These molecules tend to be used as markers and signals, as well as having some other uses. Many monosaccharides joined together make apolysaccharide. They can be joined together in one long linear chain, or they may be branched. Two of the most common polysaccharides are cellulose and glycogen, both consisting of repeating glucose monomers.
- Cellulose is made by plants and is an important structural component of their cell walls.Humans can neither manufacture nor digest it.
- Glycogen, on the other hand, is an animal carbohydrate; humans and other animals use it as a form of energy storage.
[edit]Use of carbohydrates as an energy source
- See also carbohydrate metabolism
Glucose is the major energy source in most life forms. For instance, polysaccharides are broken down into their monomers (glycogen phosphorylase removes glucose residues from glycogen). Disaccharides like lactose or sucrose are cleaved into their two component monosaccharides.
[edit]Glycolysis (anaerobic)
Glucose is mainly metabolized by a very important ten-step pathway called glycolysis, the net result of which is to break down one molecule of glucose into two molecules of pyruvate; this also produces a net two molecules of ATP, the energy currency of cells, along with two reducing equivalents in the form of converting NAD+ to NADH. This does not require oxygen; if no oxygen is available (or the cell cannot use oxygen), the NAD is restored by converting the pyruvate to lactate (lactic acid) (e. g. in humans) or to ethanol plus carbon dioxide (e. g. inyeast). Other monosaccharides like galactose and fructose can be converted into intermediates of the glycolytic pathway.
[edit]Aerobic
In aerobic cells with sufficient oxygen, like most human cells, the pyruvate is further metabolized. It is irreversibly converted to acetyl-CoA, giving off one carbon atom as the waste product carbon dioxide, generating another reducing equivalent as NADH. The two molecules acetyl-CoA (from one molecule of glucose) then enter the citric acid cycle, producing two more molecules of ATP, six more NADH molecules and two reduced (ubi)quinones (via FADH2 as enzyme-bound cofactor), and releasing the remaining carbon atoms as carbon dioxide. The produced NADH and quinol molecules then feed into the enzyme complexes of the respiratory chain, an electron transport systemtransferring the electrons ultimately to oxygen and conserving the released energy in the form of a proton gradient over a membrane (inner mitochondrial membrane in eukaryotes). Thereby, oxygen is reduced to water and the original electron acceptors NAD+ and quinone are regenerated. This is why humans breathe in oxygen and breathe out carbon dioxide. The energy released from transferring the electrons from high-energy states in NADH and quinol is conserved first as proton gradient and converted to ATP via ATP synthase. This generates an additional 28 molecules of ATP (24 from the 8 NADH + 4 from the 2 quinols), totaling to 32 molecules of ATP conserved per degraded glucose (two from glycolysis + two from the citrate cycle). It is clear that using oxygen to completely oxidize glucose provides an organism with far more energy than any oxygen-independent metabolic feature, and this is thought to be the reason why complex life appeared only after Earth's atmosphere accumulated large amounts of oxygen.
[edit]Gluconeogenesis
In vertebrates, vigorously contracting skeletal muscles (during weightlifting or sprinting, for example) do not receive enough oxygen to meet the energy demand, and so they shift to anaerobic metabolism, converting glucose to lactate. The liver regenerates the glucose, using a process called gluconeogenesis. This process is not quite the opposite of glycolysis, and actually requires three times the amount of energy gained from glycolysis (six molecules of ATP are used, compared to the two gained in glycolysis). Analogous to the above reactions, the glucose produced can then undergo glycolysis in tissues that need energy, be stored as glycogen (or starch in plants), or be converted to other monosaccharides or joined into di- or oligosaccharides. The combined pathways of glycolysis during exercise, lactate's crossing via the bloodstream to the liver, subsequent gluconeogenesis and release of glucose into the bloodstream is called the Cori cycle.

and then there was a burst of blast as of a beginning of a stein or a
‘stan’ once in the nacht of delight when the crooked engine began to
throb with an uncalculated song such as mud-fire. begin. stop. start.
we are startled by the standing silhouette as if we were the edges of
eggs. fly. you need it, you want it, and they are each and every each
of them burning, they are not screaming tho, for its not tea time. I
wanted. box. you too shall know the ill despair as if redness sang
beyond (the perceivedly habitual) commercial instability of a
substantial subjunctive or preterite habituation of wordle words and
something disguised as an aluminium south island whereas new zealand
is something about a fish and a bird or even a heteropholous of a
kylic kind that could be bright or blind or priest green in a flat of
block towers to observe a fallacy of north east west how a window had
been rushed there to the room its own believing the grey light and the
old saw in a dark dank tank where in all goodness the unconstructors
perpetually redemolish their photon ladders that gently up let them. a
strange. as if. this thus this dichte as if i knew language of whose
example: tock tuck fog bog rug mug. all this. yes. beyond. beyond
there are beamish and posilogically polished frogs that frop and flop
and slop (as if s1ip fog) whose Green is their own surprise.







Between
I
The world is shrunk to a stone:
And yet this rock has no size,
For in a crevice of it, and upon some soil,
A tree, which is minute -
Springs to hugeness
With a mass of leaf -
It quivers, penicial, throbbing.
What do you know?
What is this image
Of the grey cold stone,
Cold to my clasp -
And the rearing,
Green and yellow of the tree
As great and warm
As all its bursts of flowers?
Who is that man of gleaming grin
Who knows the white of burning bone?
What do you know?
Two ways and many have we
To know the vast world
That concentrates, now:
All the Age’s pains -
And pangs, and joys.
Children from my street
Rush from their house:
Graham in grey
Peter in blue.
II
At the Tamaki railway station
the lines shine to a convergence,
Or snake about some vague purpose.
The building, boarded up, is brown
With twenty years of trains.
There is no one here, and stains,
And red dust whirls the devil winds
Unseen unheard on the iron rails.
Clacka clack clacka clack:
You rock and roll
To death or to everything’s begin,
At the heart of Auckland - at Auckland’s heart.
But before the dreamt adventures,
Eat at the railway cafe
And drink for your journey.
You only know: the fire in treetops,
The car-flooded roads:
You are between the tree and its rock.
You are in Queen Street
There is nothing here.
Your thoughts are lead,
Your head is dead –
A man in grey is dressed in blue
He walks the pavements two by two
Counting nothing on the way
He cracks the silence with his song:
“My name is Z, I’ve been here long,
It was I did shrink this world to stone
And you my man are quite alone.”
III
The tree, the stone, send amazement thru
The vast glass eyes -
And the people stare down:
Why are the trees upside down?
Fallacious, childless man, you walk alone,
The buildings ache with stone.
A beautiful woman
Eroticates a billboard -
She is as enormous
And as stupid as Disneyland
With all its sexless eternity.
The world is truncated.
Balls of basalt roll about.
Death is forbidden. The people wait.
They know nothing.
Nobody knows nothing. We wait.
It won’t be long.
The concrete towers shiver
They are waiting
They are hoping:
A tired lady
Puts on her coat
The typewriter
Dies for fingers:
Her hands are cold -
It is 4 o’clock,
She sees the sun,
She is thinking:
“The people wait,
the people wait,
Graham and Peter
May never come.”
Beauty or desolation both? Again there is nothing left of my "past" except my memories) and the school buildings are gone.

More recent graffiti or Art. The "realist" face contrasts withe previous almost "random" work. It is almost as if the face is that of one of those idealised revolutionary figures seen in some Chinese and Soviet posters. This is a working class area so perhaps thoughts of socialism and justice are alive on the site of my old high school...who knows.

Light!

The football (rugby) field by the Tamaki Estuary. A cold but beautiful morning. Vic, my son, looks back . I can never cease my fascination with clouds and sky scenes.
.jpg)